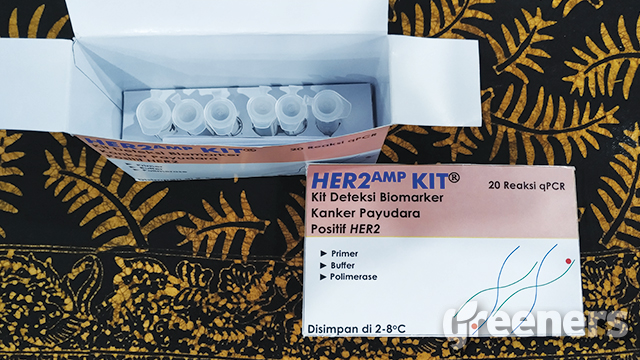
Jakarta (Greeners) – Indonesian Institute for Sciences or LIPI developed breast cancer detection kit, HER2, as the initial step to determine effective medical treatment.
“This detection technology is not the prevention but to get a sense of further steps for therapy with trastuzumab,” said Desriani, head of Biotechnology Research Center in Jakarta on Monday (04/02/2019).
The latest report released by International Agency for Research on Cancer, World Health Organization (WHO) predicts 18.1 million new cases of cancer and 9.6 millions of deaths in 2018.
Female recorded the highest number of cancer care with 2.1 millions per year in 2018, with 627,000 deaths or 15 percent.
READ ALSO: Indonesian Scientists Develops Antibiotics from the Country’s Rich Biodiversity
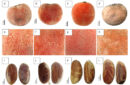
Furthermore, Desriani said that the triggers for breast cancer are three gens, — estrogen receptor, progresteron receptor, and human epidermal growth factor receptor 2 (HER2) –.
“At least 20 percent out of breast cancer patients or one out of five are positive HER2. This positive HER2 is obtained because of estrogen receptor and progresteron receptor are negative. The excess of protein of HER2 is growing cancer cells and widely spreading compare to cells with normal protein level. So, this HER2 kit will be used to detect whether patients are categorized as HER2 patients to determine the therapy for patients,” she said.
READ ALSO: Indonesia’s Drugs and Food Agency Support Herb Small Enterprises
She said that the kit is under development and still needs further tests, such as sensitivity, specification, stability, and blind test.
Ministry of Health has expressed there will be further research on the kit.
“Besides the KIT, the researchers conducted by LIPI are interferon alfa-2a non-fusion and interferon alfa-2a fusion with human serum albumin. This protein has the ability to slow down cancer cells growth,” she said.
With human serum albumin, the diagnosis via HER2 kit is validated by Chromogenic In Situ Hybridization or CISH method as 86 percent compatibility to detect the status of HER2 on cancer patients.
Reports by Dewi Purningsih